
Quick links:
Why This Matters
MK-677 has sparked widespread interest in scientific and performance research for its role in stimulating growth hormone (GH) and insulin-like growth factor 1 (IGF-1). But let’s set the record straight: MK-677 is not approved for human use anywhere in the UK, EU, or US. Its use is strictly limited to controlled, research-only environments.
In this guide, we’ll walk through:
- How MK-677 works in laboratory models
- What the research shows (and what it doesn’t)
- Potential risks flagged in studies
- Its legal classification in the UK, EU, and US
- How to handle MK-677 safely and compliantly in research settings
Let’s dig in—scientifically.
🧪 What is MK-677?
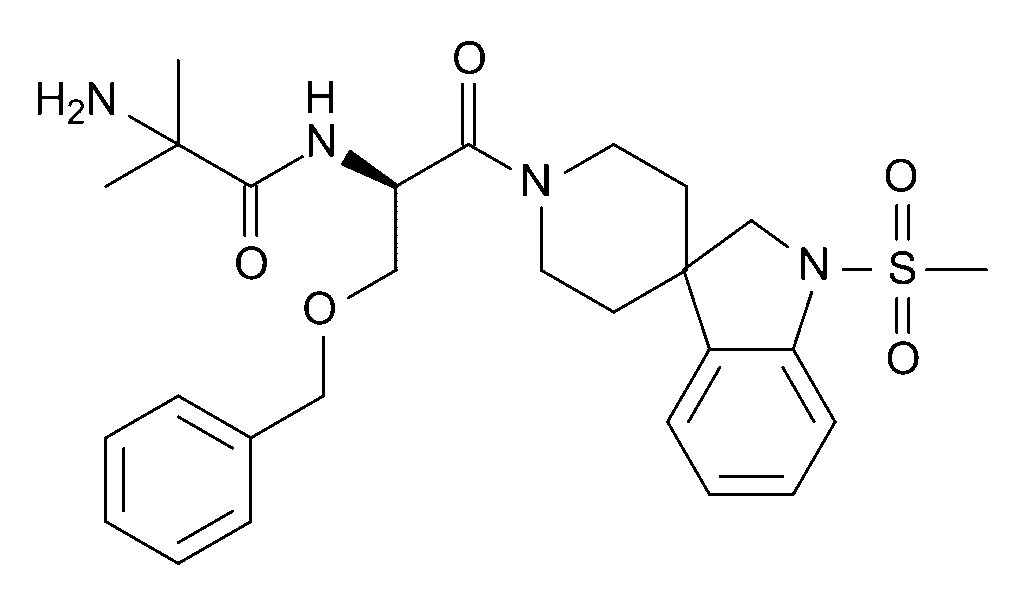
You can see above ; a substituted piperidine ring with a benzyl group and an amide-linked bulky side chain, giving it high receptor affinity and stability in plasma. Its molecular formula is C₂₇H₃₆N₄O₅S, and it exists as a mesylate salt in most research-grade preparations.”
MK‑677, also known by its development code Ibutamoren Mesylate, is a non-peptide, orally active growth hormone secretagogue (GHS). It was originally developed by Reverse Pharmacology, Inc. and later researched by Merck for its ability to stimulate the release of growth hormone (GH) and insulin-like growth factor 1 (IGF-1) without affecting cortisol levels.
It is not a SARM (Selective Androgen Receptor Modulator), not a steroid, and not an anabolic agent. Instead, MK‑677 mimics the action of ghrelin, a naturally occurring peptide hormone involved in hunger, metabolism, and GH release.
🔬 Chemical Properties
- Molecular formula: C27H36N4O5S
- CAS Number: 159752-10-0
- Molecular weight: ~528.7 g/mol
- Structure: Non-peptide small molecule
- Half-life: ~24 hours (allowing once-daily dosing in studies)
- Route of administration: Oral (capsule, solution in research)
Its oral bioavailability makes it particularly attractive for long-term endocrine and metabolic studies, compared to injectable GH or peptide GHRPs.
🧠 Mechanism of Action (MoA)
Above : The Mechanism of MK677 visualised
MK‑677 functions by binding to the GHS-R1a receptor — the same receptor activated by ghrelin, often called the “hunger hormone.” When ghrelin binds to this receptor in the hypothalamus and pituitary gland, it signals the release of endogenous growth hormone from somatotropic cells.
MK‑677 mimics this effect, with the following outcomes observed in controlled research:
| Action | Effect |
|---|---|
| GHS-R1a agonism | Increased pulsatile GH secretion |
| Hepatic GH signalling | Increased IGF-1 production |
| No suppression of natural GH | Negative feedback less pronounced than exogenous GH |
| No direct androgen activity | No affinity for AR, ER, PR, etc. |
Because of its selective receptor binding, MK‑677 does not suppress endogenous testosterone, which distinguishes it mechanistically from anabolic steroids or SARMs.
📘 In a landmark study, daily 25 mg MK-677 increased GH and IGF-1 in elderly test subjects. (Source – PMC2757071)
While originally explored for muscle-wasting and frailty, it remains unlicensed. Use outside lab settings is both unsafe and unlawful.
🧠 Because MK-677 is orally bioavailable, it’s a useful candidate in non-peptide GH research—but this same ease makes it vulnerable to misuse.
📊 What the Research on MK677 Shows

🔍 Key Research Findings on MK‑677
1. Growth Hormone (GH) & IGF‑1 Elevation
- In a landmark clinical trial of older adults (ages 60‑81), 25 mg/day of MK‑677 for 12 months increased 24‑hour mean GH by ~1.8‑fold, and IGF‑1 by ~1.5‑fold compared with baseline. PMC
- The mechanism: MK‑677 stimulates the ghrelin receptor (GHS‑R1a), triggering GH release from the pituitary, which in turn up‑regulates IGF‑1 production in the liver. PMC
Takeaway: MK‑677 reliably elevates GH/IGF‑1 in controlled human research settings.
2. Lean Body Mass (Fat‑Free Mass)
- In the same 12‑month older‑adult trial, fat‑free mass (FFM) increased by ~1.1 kg compared to a slight decline in the placebo group. PMC
- However: functional outcomes (strength, mobility, etc) did not show commensurate improvements. OUP Academic
Takeaway: While changes in body composition occur, performance gains are not well supported yet.
3. Metabolic Effects & Safety Markers
- Some metabolic side‑effects have been documented: elevated fasting glucose, reduced insulin sensitivity, increased appetite, fluid retention. OPSS flagged cardiac risk in one trial that was stopped early. OPSS
- Animal data show desensitisation in GH response over 6 weeks of administration (rats) — GH peaks returned toward baseline. PMC
Takeaway: There are meaningful safety and metabolic considerations; the long‑term risk profile remains under‑researched.
4. Functional & Clinical Benefits — Limited Evidence
- In healthy older adults, despite increased lean mass, strength and functional outcomes (e.g., walking speed, stair climb) did not improve significantly in the MK‑677 group compared to placebo. OUP Academic
- Some combination studies (e.g., MK‑677 + a SARM) have shown body mass increases, but negative effects on bone, lipids and liver markers as well. Physoc Online Library
Takeaway: Evidence for meaningful clinical or performance benefits is weak — especially in healthy populations.
🧭 What the Research Still Doesn’t Tell Us
Quality of unregulated products: Many online products labelled MK‑677 may differ in purity, dose or contain contaminants — research uses very controlled preparations.
Takeaway: Using the research to justify non‑research uses (human consumption, performance use) isn’t supported by evidence.
Long‑term safety: Data beyond 12‑24 months in healthy subjects is scarce.
Younger/athlete populations: Most human research is in older adults; extrapolation to young, healthy or athletic cohorts is speculative.
Optimal dosing, cycling & long‑term outcomes: No definitive human protocols exist.
Functional outcomes: Strength, bone health, cardiovascular benefits remain uncertain in well‑designed trials.
| Risk | Detail | Source |
| Appetite spikes | Often reported within 1–2 weeks | PMC2757071 |
| Elevated glucose | GH/IGF-1 impact on insulin sensitivity | PubMed 9467534 |
| Fluid retention | Especially in older subjects | PMC2757071 |
| Potential BMD loss | Indirectly suggested in combo studies | Wiley Physiology Reports |
Further reading : Checking the research on MK-677
⚖️ What’s the legal status of MK677?

| Region | Is MK‑677 approved for human use? | Can it be sold as a supplement? | Legal for research use? |
|---|---|---|---|
| UK | ❌ No | ❌ No | ✅ Yes (lab only) |
| EU | ❌ No | ❌ No | ✅ Yes (licensed lab) |
| USA | ❌ No (no NDA approved) | ❌ No (FDA: adulterated) | ✅ Yes (under IND or lab) |
🇬🇧 United Kingdom
Status: Unlicensed Medicine – Research Use Only
- MK‑677 is not licensed for any therapeutic indication under the Human Medicines Regulations 2012.
- It cannot be sold for human consumption.
- It is not authorised as a dietary supplement under UK law.
Official Warning:
“MK-677 is not permitted for use in food or supplements and must not be sold or marketed for human consumption.”
– OPSS Advisory on MK-677
Permitted Use:
MK‑677 may be possessed and used by qualified researchers in controlled settings (e.g., licensed research facilities or laboratories) strictly for in vitro or non-human animal studies.
🇪🇺 European Union
Status: Unapproved Investigational Compound
- The EU requires a marketing authorisation for any medicinal product under Directive 2001/83/EC.
- MK‑677 has never received EMA approval and has no MA number in any EU member state.
- It is not listed in the EU Register of Authorised Novel Foods or Supplements, and cannot be lawfully sold for human consumption.
Legal Basis:
- Directive 2001/83/EC, Article 6: No medicinal product may be placed on the market unless a marketing authorisation has been granted.
- EUR-Lex: Directive 2001/83/EC
Research Use:
MK‑677 may be used under controlled research protocols in certified laboratories that comply with national and EU safety, ethics, and documentation laws.
🇺🇸 United States
Status: Unapproved New Drug
- The Food and Drug Administration (FDA) has not approved MK‑677 for any clinical indication.
- It is classified as a “new drug” under the Federal Food, Drug, and Cosmetic Act, and may not be sold for human use without an approved New Drug Application (NDA).
Supplement Warning:
MK‑677 is sometimes sold online as a “supplement,” but this is illegal.
“Products marketed as dietary supplements that contain MK-677 are considered adulterated under the FD&C Act.”
– FDA Office of Dietary Supplement Programs
Permitted Use:
- MK‑677 can be legally obtained for research purposes by registered labs.
- It may also be used under an IND (Investigational New Drug) protocol in FDA-sanctioned human trials — but not outside of that framework.
❗️Risks of Misuse or Mislabeling

Across all jurisdictions:
- Marketing MK‑677 as a supplement, fitness aid, or “legal alternative to steroids” is unlawful.
- Importing MK‑677 for personal or clinical use may lead to seizure, fines, or enforcement.
- Websites that promote MK‑677 with dosing advice, cycle guides, or transformation claims are violating national and international law.
✅ Lawful Use (Research-Only)
To remain legally compliant, MK‑677 must be:
Stored & handled:
In accordance with chemical safety regulations and local biosafety policy.
Clearly labelled:
“For Laboratory Research Use Only. Not for Human or Veterinary Consumption.”
Sold only to:
Licensed researchers, educational institutions, or analytical testing labs.
Further reading : MK677 side effects in Research
🧷 Researcher Checklist

If you are authorised to handle MK-677, follow these essential protocols:
✅ Use in vitro or authorised in vivo (non-human) settings only
✅ Store securely, with controlled access
✅ Ensure batch documentation and COA verification
✅ Label as: “For Research Use Only – Not for Human or Veterinary Use”
✅ Comply with local and institutional biosafety rules
🧠 Pro tip: Track GH, IGF-1, glucose, and lipid biomarkers to map systemic effects in research subjects.
🧾 Summary Table
| Attribute | Value |
| Class | Growth hormone secretagogue |
| Structure | Non-peptide, orally bioavailable |
| Status | Research use only – unlicensed in UK/EU/US |
| Legal use | In vitro or approved animal models only |
| Risks | Appetite, glucose intolerance, fluid retention, possible BMD reduction |
Summary & Downloadable Resources for MK677
| Download the MK677 Research checklist here |
| MK677 White Paper Dossier |
| Compliance datasheet |
| MK677 Lab safety poster |
| Dosage clinical trial overview |
MK-677 remains an important tool for endocrine and metabolic research, but its unlicensed status demands caution. If you’re working with it in any capacity:
- Know the laws
- Follow protocols
- Never extrapolate research findings to human application without regulatory approval
“Treat investigational compounds with the respect they demand—because pushing boundaries starts with protecting safety.”
FAQ & Quick reference
🔍 “What is MK-677 and what does it do?”
MK-677, also known as Ibutamoren, is a non-peptide growth hormone secretagogue. It mimics ghrelin by binding to the GHS-R1a receptor, triggering increased secretion of growth hormone (GH) and insulin-like growth factor 1 (IGF-1).
In controlled research studies, MK-677 has been shown to:
- Elevate GH and IGF-1 levels
- Promote nitrogen retention
- Modulate sleep cycles
- Increase fat-free mass in older adults (without improving strength)
It is strictly for non-human laboratory use.
⚖️ “Is MK-677 legal in the UK / USA / Europe?”
No – not for human use.
MK-677 is not licensed or approved in the UK, US, or EU. It:
- Cannot be sold as a supplement
- Cannot be marketed for human or veterinary use
- Can only be supplied for authorised laboratory research
See regulators:
💊 “Can I take MK-677 for muscle growth or sleep?”
Absolutely not. MK-677 is not approved for ingestion or therapeutic use.
Although early studies showed increased GH, IGF-1 and lean mass, MK-677 is:
- Unlicensed
- Unapproved for human trials without IND/clinical oversight
- Not tested for safety in healthy users long-term
Its only legal use is for in vitro or non-human models in controlled lab environments.
⚠️ “Is MK-677 dangerous?”
In research settings, the following effects have been observed:
- Increased appetite
- Elevated blood glucose and insulin resistance
- Water retention and oedema
- Fatigue or lethargy
- Bone density changes (in long-term studies)
These effects were seen under medical supervision in clinical trials. Use without regulation is considered high risk and potentially unlawful.
🧪 “What’s the half-life and dosing of MK-677?”
In published studies:
- Half-life: ~24 hours
- Dose used: 25 mg/day in elderly subjects (Walker et al., 1998)
But again, this information is for reference in designing experiments only. MK-677 is not approved for human dosing, and any reference to cycles or regimens outside of peer-reviewed research is non-compliant.
🧾 “Is MK-677 a SARM or a steroid?”
No.
MK-677 is a ghrelin mimetic, not a SARM and not a steroid. It:
- Has no androgenic activity
- Does not bind the androgen receptor
- Works independently of testosterone or anabolic pathways
It’s often mistaken for SARMs due to overlap in online forums — but in research terms, it belongs to a completely different pharmacological class.
🧬 “Can I buy MK-677 legally?”
Yes — if you are a qualified researcher purchasing from a legitimate chemical supplier. The product must:
- Be labelled “For Research Use Only – Not for Human or Veterinary Use”
- Include full certificate of analysis (CoA) and safety documentation
- Be stored and handled under biosafety protocols
Consumer sale, import for self-use, or retail “supplement” marketing is illegal in the UK, EU, and US.
🔐 “Where is MK-677 used in science?”
Common areas of preclinical research include:
- Age-related GH decline and frailty models
- Cachexia (muscle wasting)
- GH/IGF-1 endocrine pathway studies
- Sleep and circadian rhythm modelling
- Metabolic disease (insulin resistance, obesity)
All studies must be conducted under appropriate ethical review and regulatory approval.
🧷 “How should MK-677 be stored for research?”
Standard lab handling instructions:
- Store at –20°C (long-term) or 2–8°C (short-term)
- Avoid exposure to heat, light, moisture
- Use gloves and PPE
- Track batch, expiry, and usage logs
- Dispose of via licensed chemical waste protocols
Further guidance : SARMs storage
📚 “Where can I read more real studies on MK-677?”
Here are high-authority sources:
- Walker et al. 1998 – GH/IGF response in elderly
- Nass et al. – Long-term MK-677 in older adults (PMC)
- Ghrelin receptor agonists in endocrine research (PMC)
